There are known constitutional mutations associated with bone marrow failure and progression to malignancy, including myelodysplastic syndrome (MDS) and acute myeloid leukemia (AML). For the majority of the defects associated with clonal hematopoiesis and progression to MDS/AML, there are no specific treatments. We sought to determine if messenger RNA (mRNA) transfer can restore expression of the normal proteins, modulate abnormal proliferation and differentiation, and enhance response to chemotherapy and targeted agents.
CCAAT enhancer-binding protein alpha (CEBPA) is a transcription factor involved in myeloid differentiation that is mutated in ~7-20% of patients with AML (Su L et al., Frontier Oncol. 2022). A germline alteration was identified in 4.5% of AML patients with CEBPA mutation (Ho PA et al., Blood 2009). While it was originally believed that biallelic mutations of CEBPA confer favorable prognosis, more recently it was realized that a single basic region leucine zipper (bZIP) mutation in the C terminal region suffices to confer favorable risk, as categorized by ELN2022. There are currently no targeted therapies for mutated CEBPA, and no known specific therapy for patients with constitutional or acquired CEBPA mutations. Although most patients with mutated CEBPA achieve initial CR, there is a moderate relapse rate (30-50%).
The objective of this study was to determine if restoration of normal CEBPA mRNA would result in expression of normal CEBPA protein in AML cell lines (e.g.-U937), as well as an AML cell line with mutated CEBPA (KO52), and in primary AML blasts, including those with mutated CEBPA. We tested whether expression of normal CEBPA would lead to phenotypic changes including overcoming differentiation block, induce apoptosis, and eliminate leukemic blasts. There are normally two translation initiation sites, leading to production of the CEBPA-p42 or CEBPA-p30 proteins. U937 has high level expression of normal CEBPA-p42, while the KO52 cell line has two CEBPA mutations, p.E50* and p.L324P, the former leading to decrease in CEBPA-p30.
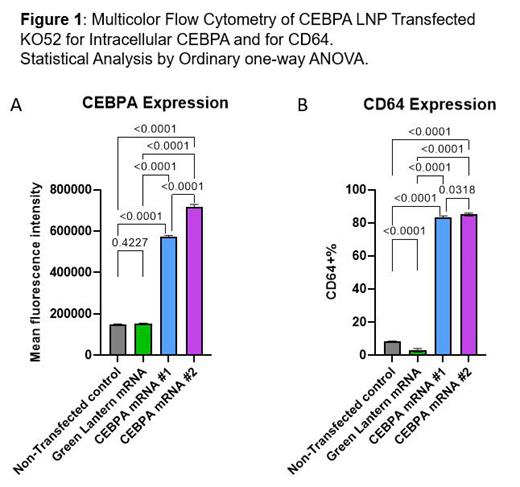
To study the consequences of mRNA transfer, we performed immunofluorescence, multicolor flow cytometry (MFC), and Western blot (WB) analysis for non-transfected control cells, GreenLantern mRNA lipid nanoparticle (LNP) transfected control cells, and cells transfected with two types of CEBPA mRNA LNP (#1 and #2). We observed transfection efficiencies of 90-97% in cell lines and 20-95% in primary cells. Figure 1A shows the mean fluorescence intensity (MFI) of Alexa Fluor® 647-labeled monoclonal antibody to the C-terminus of CEBPA. Wild-type CEBPA-p42 was expressed in U937 and KO52 cell lines at 2-4 times control levels by WB quantitation. With two additions of LNP at 0h and 48h, the induced CEBPA protein maintained stable expression up to 5 days. Immunofluorescence demonstrated staining of CEBPA in the nucleus, co-localizing with nuclear DAPI fluorescence.
To evaluate the functional consequences of CEBPA introduction, we analyzed morphology by Wright/Giemsa staining, proliferation by cell counting, apoptosis by Annexin V staining, cell cycle using BrdU labeling assay, and immunophenotype with stem and lineage markers including CD45, CD34, CD38, CD123, CD117, HLA-DR, CD13, CD33, CD16, CD11b, CD64, CD14. Strikingly, the enhanced expression of CEBPA was associated with decrease in proliferation and fraction in S phase, and increase in apoptosis and myeloid differentiation, suggesting induction of differentiation that could have therapeutic implications similar to ATRA in acute promyelocytic leukemia. The myeloid differentiation was evident by MFC showing decreased expression of primitive markers CD34, CD117 and HLA-DR, with increased expression of CD64 (Figure 1B), CD38, and CD11b. With this method we have also transfected primary AML patient blasts including a sample that harbored 4 CEBPA mutations. We observed 95% transfection efficiency and >50% decrease in cell number relative to controls with increased myeloid differentiation. Studies of the combination of CEBPA mRNA LNP transfection with targeted inhibitors to the other mutations present in this sample are in progress.
Our findings reveal a novel therapeutic strategy using CEBPA mRNA LNP applicable to inherited or acquired disorders of CEBPA. This approach of restoration of normal mRNA could have potential for therapy of many pre-malignant and malignant disorders.
Disclosures
Kfoury:Moderna, Inc: Current Employment. Hoggatt:Moderna, Inc: Current Employment. Marcucci:Ostentus Therapeutics: Current equity holder in private company, Research Funding. Becker:Accordant Health Services: Membership on an entity's Board of Directors or advisory committees; GPCR Therapeutics: Research Funding; Glycomimetics: Research Funding; Pfizer: Research Funding.